Lab Equipment Scientific research equipment:
 | STA 449C Jupiter+QMS 403C Aeolos Netzsch Simultaneous thermogravimetry + differential scanning calorimetry + mass spectrometry technique (three techniques in one device Temperature range: room temperature – 1500?C; - Heating and cooling rates: 0.01 K/min – 50 K/min; - Mass of sample: 1 – 5000 mg; - Atmosphere: inert, oxidative, static, replacement, dynamic; - Simultaneous thermal experiments with mass-spectrometry analysis are available; - Equipped with a unique system PulseTA? The values for measuring : heat, heat effect (heat, enthalpy) and the melting temperature, crystallization, recrystallization, phase transitions, glass transition, chemical reactions, thermal decomposition temperature, weight loss, residual mass, the oxidation temperature. The presence of the mass spectrometer allows the analysis of gas - thermal analysis products. |
 | Accelerating Rate Calorimeter ARC? 254 (Adiabatic) This equipment is used for physical-chemical studies of oxidation and pyrolysis of crude oils and bitumen as well as behavior of explosive and thermally unstable compounds. This equipment has special VariPhi module, that enables to study reactions in different modes, and an injection system for input of liquid and gaseous compounds during the experiments. Heating rate: 0.01 - 10 K/min Temperature range: 0-550˚С Pressure range: 0-600 bars Sample volume: 0.,5 – 130 mlreactionary calorimetry) |
 | Ramped Temperature Oxidation (RTO) (Plant for Oil Oxidation Studies under Linear Temperature |
 | DSC 214 Polyma Netzsch Designed for studying the thermal effects of phase transitions, chemical reactions, pseudo crystalline transitions, including at low temperatures. Technical Key Data for the DSC 214 Polyma 1) Temperature range - 170 ÷ 600?C 2) Heating/Cooling rate – 0.001 K/min to 500 K/min* 3) Indium Response Ratio > 100 mW/K ** 4) Resolution (technical) 0.1 ?W 5) Enthalpy precision – ?0.1% for indium; ?0.05% to ? 0.2% for most samples 6) Specific heat determination – Optional 7) Temperature modulation – Optional 8) Cooling device options a) Compressed air cooling (RT to 600?C) b) IC40 (-40?C to 600?C) c) IC70 (-70?C to 600?C) d) LN2, automatically controlled (-170?C to 600?C) |
 | High-Pressure Technical Data 1) Wide pressure range: vacuum ... 15 MPa (2140 psi) 2) Exact pressure adjustment 3) Pressure accuracy ? 0,075 MPa 4) Temperature range: -150?C to 600?C (Temperature range is pressure and gas type dependent: ? -150?C to 600?C (at 1 bar) ? -90?C to 600?C (at 50 bar) ? -50?C to 450?C (at 150 bar)) 5) Heating rates: 0.01 K/min to 100 K/min 6) Measurements in different atmospheres inert (N2, He, Ar) ? reducing (H2) ? oxidizing (O2, air) ? other gas types on request 7) Exact gas flow regulation (up to 500 ml/min) 8) Exchangeable sensor plate 9) Allows measurements in according with ASTM D5483, D6186, E1782, E1858, E2009 10) Evaluation of oxidative-induction time (OIT) included Differential Scanning Calorimetry |
 | Thermo-Microbalance TG 209 F1 Libra Netzsch A thermobalance is used to study the thermal stability, oxidation and pyrolysis, kinetics of chemical reactions Technical Key Data: 1) Temperature range 10?C - 1100?C 2) Heating and cooling rates 0.001 - 200 K/min 3) Cooling time (1100?C to 100?C) ∼ 12 min 4) Measuring range 2000 mg 5) Resolution 0.1 ?g 6) The integrated gas supply unit with mass flow controllers (MFCs) accommodates up to two purge gases and one protective gas. |
 | Microcalorimeter microDSC7 evo Setaram Microcalorimeter is used to study heat capacities and description of thermal effects of phase transition and chemical reactions. Technical Key Data: 1) Temperature range -45?C – 120?C 2) Programmable temperature scanning rate (heating and cooling) 0.001 to 2 C.min-1 3) Cells 1 ml |
 | Oxygen bomb calorimeter IKA 6000 Calorimeter is used to study thermal effects of combustion and formation of substances as well as calorific value of different materials. Technical Key Data: 1) Temperature range 20?C – 30?C 2) Scanning mode: adiabatic, isoperibol, dynamic 3) Automatic ignition |
 | Rheometer MCR 302 Anton Paar Rheometer is used to study rheological properties of low and high-viscos substances, oil, bitumen, petroleum, paving asphalt, resins, polymer solutions, gelling agents. Technical Key Data: 1) Temperature range -40?C – 400?C 2) Pressure: 1-300 atm 3) Modes: rotational and oscillatory 4) Normal force range 0.01-50 (+0.005) 5) Shear stress measurement in 2 modes: – shear velocity control; – shear stress control. |
 | SVM? 3001 Stabinger Viscometer Anton Paar Viscometer is used to study the kinematic and dynamic viscosity and density within the range of temperature. Technical Key Data: 1) Dynamic viscosity (mPa.s): 0,5 – 20,0002) Density (g/cm3): 0,65 – 3,00 3) Temperature range: 15 – 105 0С |
 | Automated system Accelerated Surface Area and Porosimetry (ASAP) 2020MP (Micromeritics) Automated system ASAP 2020MP (Micromeritics) is used to analyze the surface area and pore structure study of dispersed materials. Technical Key Data: 1) Definition of specific surface area 0,01 m2/g2) Definition of pore size distridution (nm): 0,35 – 500 in diameter |
  | Series 4560 Mini Reactors |
 | Particle size and adsorbtion potential analyzer Matec  |
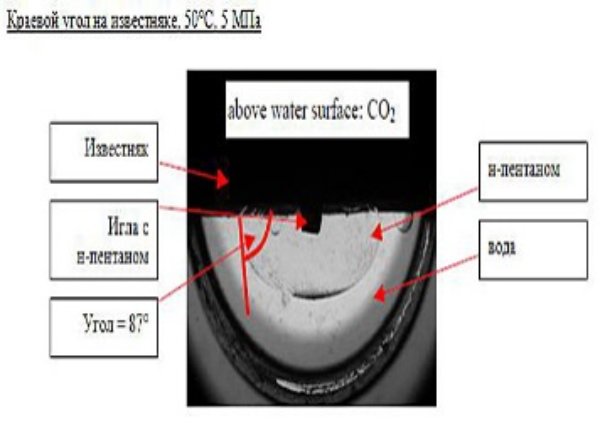
 | Drop Shape Analyzer – DSA100HP Analyzer is used to define surface/interface tension and wetting contact edge under the pressure up to 70 MPa and visually observe the behavior of materials under these conditions. Solidification and swelling of the sample may provide additional information about the properties of the system.
|